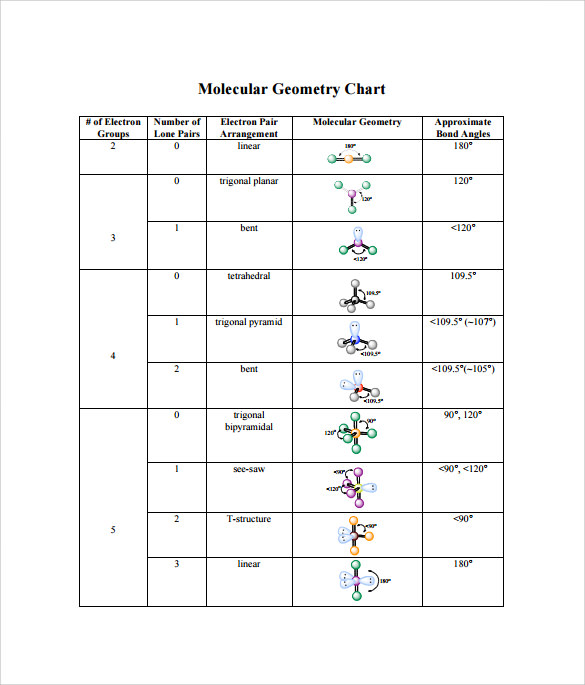
Um So if we take a look here, we place the least electro negative element in the center. So for this example, it says determined the electron geometry for the following molecule CH two. But there are rules to draw the best structure. Recall there are many possible lewis dot structures that exist. Remember in the case of electron geometry, lone pairs on the central element and surrounding elements are treated as the same Hide transcripts If it's hard for you to remember a particular electron geometry name and you'll be able to get through this. So just remember some of these memory tools. So remember if you have six electron groups, your electron geometry is octahedron. So eight tentacles -2, -2 gives me six six electron groups. So an octopus has eight tentacles right or eight legs And it has two months. Well we can play on this idea of optimizing eight. But here we're talking about six electron groups. This one is gonna require a little bit more imagination in terms of remembering it Octa you might say hey jules, octo means eight. So again that could be a combination of all surrounding elements That add up to six or some combination of surrounding elements and long pairs.  
Alright, finally we have six electron groups. How am I supposed to remember this name? Well we know that it has five electron groups and we're gonna say your try means three by here means to three plus two will give me five. Here we have two surrounding elements in three lone pairs. Right so here we have three surrounding elements. Which could be all surrounding elements or a combination of surrounding elements and lone pairs. 
So the central element has five electron groups around it. Now the next two Let's look at the next two We have five electron groups for the next one. The electron geometry name would be tetra hydro tetra means four for electron groups. And remember that could just be different situations where they're all surrounding alliance or their surrounding elements with a mixture of lone pairs as long as it adds up to four electron groups. Well we're gonna say we have three electron groups and it starts with tri tri meaning three. Just make sure you're spelling it correctly and how to remember this. Now the beauty of chemistry is that we can pronounce things differently, but all that really matters is that you get the spelling correct? Okay, so you might hear trickle planner, Trigano Planner, Tribunal planner Trigano Planner. The electron geometry name would be tribunal plainer. Remember we're treating lone pairs and surrounding elements as the same in this case. And remember these electronic groups could either be three surrounding elements Or there could be two surrounding elements and one lone pair. So again, we have carbon in the center and we have two electron groups. Okay, so line for linear and two points because there's two electron groups. Is that recall that there are two points in a straight line. The geometry here the electron geometry would be called linear. So that's why it has two electron groups. Here we have carbon dioxide as as an example, carbon is a central element. And let's say that our molecule has two electron groups. So here we're gonna start out with the first one. So if we take a look here at this chart, we're breaking things down in terms of number of electron groups, the orbital shapes where we just give certain examples that fit this particular row, the electron geometry name and then a memory told to help us to remember that electron geometry name. And when it comes to the electron geometry, we're gonna treat lone pairs and surrounding elements as the same. So when we say electron geometry realize that it represents the simplest system for geometrical shapes that focuses on the number of electron groups around the central element.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
